radioactivity
radioactive decay
radioactivity is the spontaneous and random emission of radiation from unstable atomic nuclei.
types of radiation
alpha radiation
- alpha particles consist of two protons and two neutrons
- they have a positive charge
- very strongly ionising
- low penetrating power
- stopped by paper or a few cm of air
beta radiation
- beta particles are fast-moving electrons
- they have a negative charge
- moderately ionising
- moderate penetrating power
- stopped by a few mm of aluminium
gamma radiation
- gamma rays are electromagnetic waves
- they have no mass and no charge
- weakly ionising
- very high penetrating power
- reduced by thick lead or concrete
deflection in fields
in an electric field:
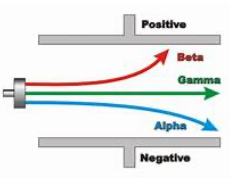
in a magnetic field:
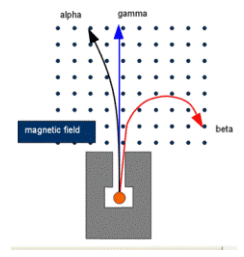
you can use the left hand rule here.
beta deflects more than alpha since it's lighter, meaning the same force causes greater deflection.
ionisation
ionisation occurs when radiation removes electrons from atoms, forming ions.
- alpha is the most ionising
- gamma is the least ionising
random nature of decay
radioactive decay is random and cannot be predicted for a single nucleus.
- the decay rate of a large number of nuclei is predictable
- external factors do not affect decay rate
dangers and safety
- ionising radiation can damage living cells
- high doses may cause mutations or cancer
- sources should be handled with shielding and distance
half-life
half-life is the time taken for the count rate (or number of undecayed nuclei) of a radioactive substance to fall to half its original value. after each half-life, the count rate halves.
the relationship between count rate and half-life can be written as:
final count rate = initial count rate / 2ⁿ
where n is the number of half-lives that have passed.
for example, if the initial count rate is 400 counts per second: after 1 half-life → 200, after 2 half-lives → 100, after 3 half-lives → 50.
make sure to remove and add the background count rate again when calculating half-life.